Onychodystrophy Testing
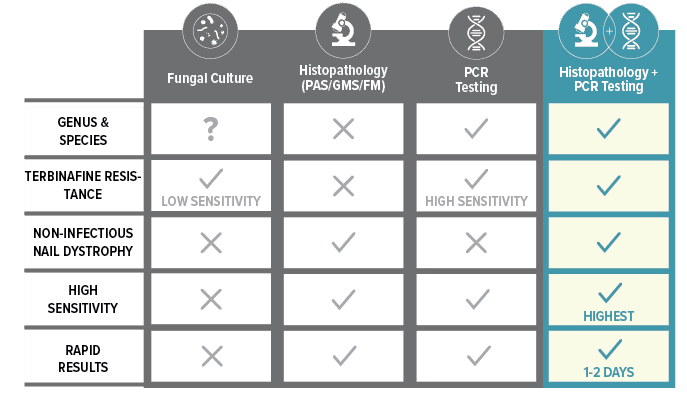
Histopathology + PCR = The Complete Picture
Various nail disorders appear identical and clinical examination alone may not be enough.
Definitive Diagnosis
Laboratory testing identifies the underlying cause of nail dystrophy – infectious, non-infectious, neoplastic processes, and trauma
Complete Picture
Histopathology + PCR Testing provide comprehensive information to treat nail dystrophy effectively.
Targeted Treatment
Identifying the genus and species of the infectious organism allows you to target therapy with precision.
Complete the form below and your local sales account manager will reach out to answer your questions about BakoDx histopathology + PCR onychodystrophy testing.
The Complete Picture
*When used in combination; Bako Nail PCR Testing: 99.9% Analytical Specificity, 86% Clinical Sensitivity. Bako’s PCR testing highly correlates with histopathology. Histopathology: 93% Clinical Sensitivity, 96% Clinical Specificity. Fungal culture: 42% False Negative Rates
Histopathology
Microscopic evaluation of a specimen to determine infectious or non-infectious pathologies including traumatic, inflammatory or neoplastic processes.
Periodic Acid–Schiff (PAS): Identifies non-degenerated fungal elements
Grocott Methenamine Silver (GMS): Identifies degenerated fungal elements
Fontana-Masson (FM): Identifies pigmented saprophytes and melanin pigment which may be associated with a melanocytic neoplasm
PCR
Molecular analysis of a specimen to determine if DNA of an infectious organism is present.
Onychodystrophy PCR Test: Identifies genus and species of most common fungi (dermatophytes, saprophytes, and yeasts) as well as the bacteria, pseudomonas aeruginosa, that cause nail dystrophy
PCR with Terbinafine Resistance Reflex: When positive for T. rubrum or T. mentagrophytes, detects mutations associated with terbinafine resistance
Educational Resources for Onychodystrophy
Disclaimer: BakoDx, as a partner to your practice, provides learning resources to help you better serve your patients. The information contained within this document should be used at a physician’s discretion. BakoDx assumes no liability for its content.